First-line treatment of systemic juvenile idiopathic arthritis (JIA) with anakinra (Kineret) was highly effective as monotherapy, minimizing the need for glucocorticoids, a single-center prospective study found.
Among 42 patients treated early with the recombinant interleukin (IL)-1 receptor antagonist anakinra who had not responded to treatment with nonsteroidal anti-inflammatory drugs (NSAIDs), 76% had inactive disease at 1 year and 52% had inactive disease off the medication, according to Sebastiaan J. Vastert, MD, PhD, of University Medical Center Utrecht in the Netherlands, and colleagues.
And only one-third of patients required glucocorticoids, compared with 80-90% of patients in previous cohorts, the researchers reported online in Arthritis & Rheumatology.
“The first-line initiation of biological therapy is a paradigm shift compared to conventional step-up regimens used in systemic JIA,” Vastert and colleagues stated.
“The group from Utrecht present compelling data about the effectiveness of IL-1 inhibitor monotherapy in this cohort of patients newly diagnosed with systemic JIA that was refractory to NSAIDs,” observed Timothy Beukelman, MD, of the University of Alabama at Birmingham, who was not involved in the study.
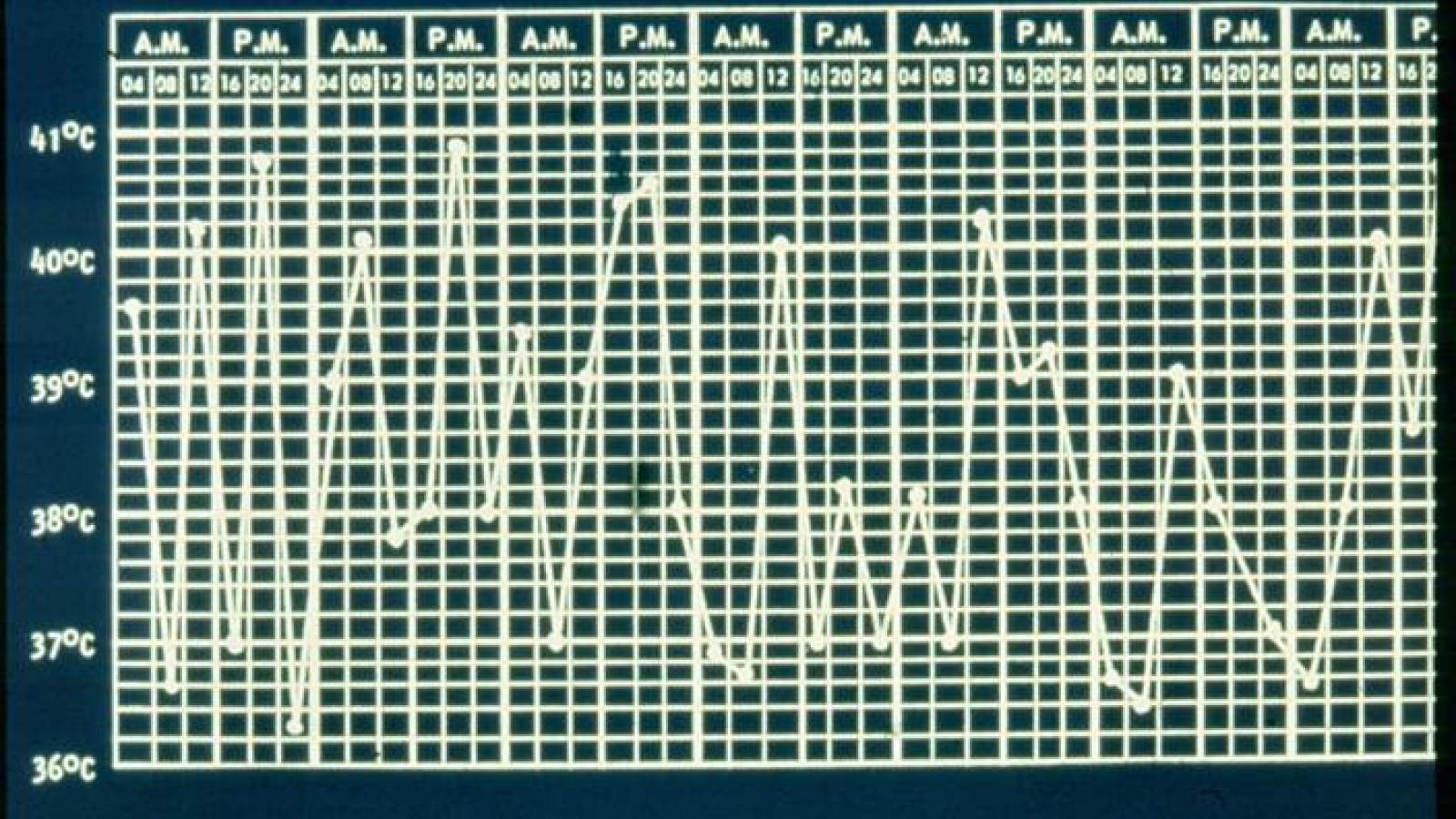
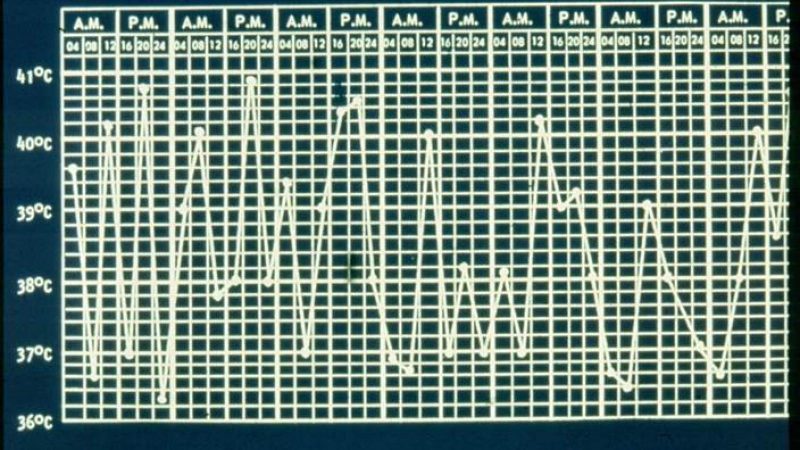
Systemic JIA differs from other forms of JIA in that it is characterized by spiking quotidian fevers, rash, hepatosplenomegaly, and elevated erythrocyte sedimentation rate or C-reactive protein, with or without arthritis at disease onset. The disease course varies considerably, with some patients having a monocyclic course.
Disease-modifying antirheumatic drugs (DMARDs) that have been helpful in other JIA subtypes have not provided relief in the systemic subtype, and treatment has often relied on long-term steroids, with their numerous adverse effects including growth restriction and obesity.
The discovery of the involvement of the IL-1 pathway in the pathogenesis of systemic JIA, first reported about 15 years ago, opened new options in treatment, and previous studies targeting biologic pathways have demonstrated efficacy. However, in earlier studies, biologic agents usually were given in combination with DMARDs and/or glucocorticoids and as a second- or third-line agent.
Further research revealed that the IL-1 activation was most prominent in the early phase of disease, suggesting the possibility of an early window of opportunity for IL-1 blockade. Accordingly, the Utrecht group began enrolling patients in 2008 using a treat-to-target approach, initiating anakinra treatment if symptoms persisted for 7 days on indomethacin or other NSAIDs.
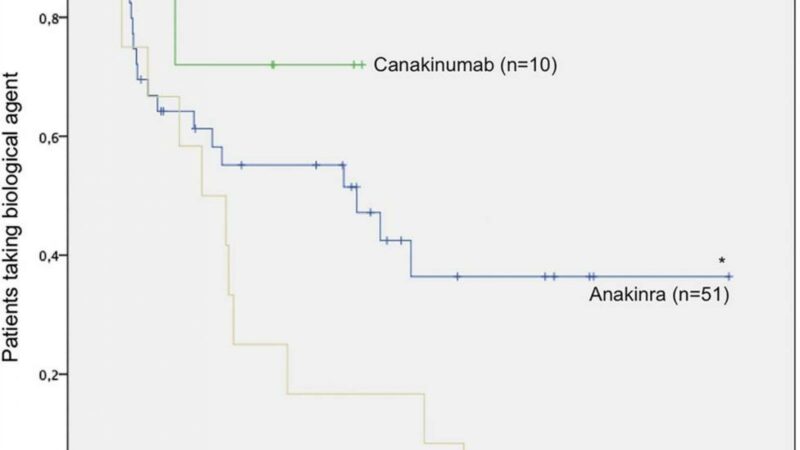
The medication was started in dosages of 2 mg/kg/day, and if fever persisted for 3 days, the dosage was increased to 4 mg/kg/day. Prednisolone could be added if symptoms continued, or the patient could be switched to other biologics such as canakinumab (Ilaris) or tocilizumab (Actemra).
The cohort included 42 patients whose mean age at symptom onset was 7 years. The majority were boys, and 12 had the systemic disease manifestations without arthritis at baseline.
After only 1 month of treatment, 55% of patients had completely inactive disease with anakinra monotherapy, and only two had used prednisolone. After 3 months, 83% had inactive disease without using steroids, and the median time to reach inactive disease was 33 days.
Tapering the medication was considered important, as anakinra is given in daily subcutaneous injections, which can be burdensome for pediatric patients. After a mean of 3.7 months of treatment, 33 patients began tapering and 31 were able to stop the drug; median duration of treatment was 6.1 months. However, half of patients had recurrences while tapering, so “future studies should try to identify and/or validate biomarkers of subclinical disease activity in order to optimize taper and stop strategies,” the researchers noted.
A total of 35 patients were followed for 3 years and 25 for 5. Among those followed for 5 years, all had inactive disease and 15 were drug-free, with two remaining on anakinra, two having switched to canakinumab and one to tocilizumab, and one using an NSAID and intra-articular steroids.
One patient who had very severe disease that had not responded to anakinra, methotrexate, canakinumab, tocilizumab, or glucocorticoids died from macrophage activation syndrome. Three others also experienced that serious complication, with two possibly having been triggered by viral infections. Fewer than 5% of patients reported any disease damage.
Factors that were predictive of inactive disease at 1 year were early response, meaning inactive disease after 1 month of treatment (OR 27, 95% CI 2.92-47.5, P=0.003), and a high neutrophil count prior to initiation of treatment (OR 1.19, 95% CI 1.05-1.42, P=0.021), with the optimal cutoff a count of 9×109/L.
“Taken together, our data demonstrate that a treat-to-target strategy using first-line monotherapy with a recombinant IL-1 receptor antagonist is a highly effective treatment strategy for systemic JIA patients, both for short- and long-term outcomes,” Vastert and colleagues said.
Beukelman agreed: “I personally endorse this treatment approach of early use of IL-1 inhibition,” he told MedPage Today.
“While it is true that many patients with systemic arthritis may have a self-limited disease course, we cannot currently identify these patients at initial presentation,” said Beukelman, who has written extensively about this topic. “The risks of a several-months course of anakinra appear small compared to the benefits of rapid disease control, which may include a decreased incidence of subsequent destructive polyarthritis as observed in this cohort.”
Limitations of the study, the researchers said, included its single-center design and lack of a control group.
The authors reported financial relationships with Novartis, Sobi, ZonMw, Pfizer, and AbbVie.
Beukelman reported financial support from Sobi.
Author: John Swope
Related Content
-
November 26, 2019
A disease you’ve never heard of is becoming increasingly common and carries a…
-
August 8, 2019
A single-center cohort analysis shows that lung disease (LD) is increasingly seen in…
-
January 17, 2018
Since 2000, the German Biologics register (BiKeR) has prospectively enrolled children with juvenile…
-
April 5, 2018
The German Autoinflammatory Disease (AID) registry has studied the effects of the IL-6…
-
May 5, 2015
A retrospective review of 77 SoJIA patients revealed that 50-70% achieved inactive disease or remission…
-
November 9, 2021